INTRODUCTION
Some of the most consequential challenges reported among people living with HIV (PLHIV) are socially mediated, including stigma1 and may be differently perceived among men who have sex with men (MSM), men who have sex with women (MSW), and women, depending on social norms. For example, MSW have high co-prevalence of other stigmatizing conditions such as substance use disorder, particularly injection drug use2. MSM may experience significant social stress at the intersection of their identities as PLHIV and MSM, especially in settings where being gay is criminalized or tabooed3. The psychosocial and emotional challenges faced by women living with HIV are multifaceted as women are more likely to be the caregivers, less educated, financially dependent, and at higher risk of interpartner violence4-7.
Understanding differences in perceived treatment needs by gender/sexual orientation is critical to tailoring treatment for better health outcomes. Solely exploring men–women differences, while important, may however mask underlying differences between MSM and MSW. This is a key concern because perceived treatment needs of MSW remain less understood, despite this group being the third largest population living with HIV, including many vulnerable groups such as people who inject drugs8. To fill this gap in knowledge, we performed comparative analyses among MSM, MSW, and women, using self-reported data for 2389 PLHIV in 25 middle- and high-income countries. We have previously reported some findings from these data in relation to polypharmacy, treatment adherence, patient–provider engagement, and aspirations towards improved HIV treatment9-13. In this work, however, we explore some of these themes from the perspective of disparities and health equity. Two fundamental questions were of interest. 1) What is the magnitude of the disparity in health-related outcomes by gender/sexual orientation? 2) Is the association between behavioral interventions (e.g., counseling about ‘Undetectable Equals Untransmittable’) and health-related outcomes different among MSM, MSW, and women? Elucidating these issues can inform patient care as well as public health decision-making regarding research, patient education, and healthcare priorities to help eliminate inequalities.
METHODS
Study population and sampling approach
This study, the 2019 Positive Perspectives (Wave 2), was from an online convenience sample of PLHIV living in 25 middle- and high-income countries. The first wave, conducted in 2017, comprised only high-income countries; the second wave was expanded to include 6 middle- and 19 high-income countries. Participating countries in the second wave, and sample sizes, were as follows: USA (n=400), South Africa (n=179), Russia (n=150), United Kingdom (n=123), Australia (n=120), Canada (n=120), France (n=120), Germany (n=120), Italy (n=120), Spain (n=120), Japan (n=75), Mexico (n=63), Portugal (n=60), Brazil (n=58), Switzerland (n=55), Taiwan (n=55), Netherlands (n=51), Argentina (n=50), Austria (n=50), Chile (n=50), China (n=50), Ireland (n=50), Belgium (n=50), Poland (n=50), and South Korea (n=50). Inclusion criteria for all participants were: 1) consenting adults aged 18–84 years, and 2) self-identified as been diagnosed with HIV and on antiretroviral treatment (ART) at the time of the survey. Of 7177 individuals screened during the wave 2 survey, 4203 were eligible, and 2389 completed the questionnaires (overall response rate: 57%). Ethical review was provided by the Pearl Institutional Review Board (#18–080622, covered all 25 countries) and the Sefako Makgatho Research Ethics Committee (for South Africa specifically, #SMUREC/M/223/2019).
Measures
Gender/sexual orientation status
Designation as MSM (n=1018), MSW (n=479), or women (regardless of sexual orientation, n=696), was derived from two separate variables for self-classified gender and sexual orientation. Individuals who identified their gender as ‘Man (including transman)’, and their sexual orientation as ‘Homosexual/Gay/Lesbian’ were classified as MSM. Individuals who identified their gender as ‘Man (including transman)’ and their sexual orientation as ‘Heterosexual/Straight’ were classified as MSW.
Health-related outcomes/experiences
Self-reported virologic control was defined as a response of ‘undetectable/suppressed’ to the question: ‘What is your most recent viral load?’. Polypharmacy was defined as taking ≥5 pills/day (for HIV or non-HIV conditions), or currently taking medicines for ≥5 medical conditions, including HIV9. Suboptimal adherence was a report of ≥1 reason for which the individual missed HIV medication ≥5 times within the past month10. Data were also collected on willingness to share HIV status, and reasons for not sharing HIV status with others in the past. The survey further asked participants what issues they prioritized the most when they first started their HIV treatment, and what their current priorities were. Self-rated health was deemed optimal if self-classified as ‘Good’/‘Very good’. In addition, respondents provided information on various aspects of communication with their healthcare providers (HCPs), including whether ‘my provider has told me about “Undetectable = Untransmittable” (U=U)’ and whether they felt comfortable discussing with their HCP ‘concerns about the safety of others/preventing transmission’. Various barriers to discussing salient health issues with HCPs were assessed using the question: ‘Why, if at all, would you feel uncomfortable raising concerns with your main HIV care provider?’. Individuals with a response of ‘None – I would always be comfortable’, were classified as perceiving no barriers in discussing salient health issues with their HCP.
Statistical analyses
Prevalence estimates were calculated overall and by gender/sexual orientation within middle- and high-income countries separately (World Bank classification using per capita income)14. Comparisons of prevalence estimates were done with 2 tests (p≤0.05). Trends in self-reported diagnosis of various conditions by time since HIV diagnosis were explored using joinpoint and age-adjusted logistic regression. For each line segment in the joinpoint regression, we computed annual percentage change (APC) with 95% confidence intervals; estimates of average annual percentage change (AAPC) were used to summarize overall trends across all segments. We further used multivariable logistic regression to examine whether MSW-MSM and MSW-women differences remained significant after adjusting for HIV duration, underlying comorbidities, and various indicators of healthcare access. To determine whether the magnitude of the association between exposure to HCPprovided information about ‘Undetectable = Untransmittable’ and health-related outcomes differed by gender/sexual orientation, separate logistic regression analyses were performed among MSM, MSW, and women, each adjusting for education, employment, home ownership, age, year of HIV diagnosis, and country-specific human development index – a composite measure of life expectancy, education, and per capita income. Statistical analyses were with R Version 3.6.1.
RESULTS
Pooled data from middle- and high-income countries showed that mean (SD) age was 43.4 (12.2) years among MSM, 36.6 (10.6) among MSW, and 41.0 (11.9) among women. The percentage with >high school education was: MSM (80.1% [815/1018]), MSW (70.8% [339/479]), and women (70.1% [488/696]). The percentage who rented/owned their own housing was: MSM (78.0% [794/1018]), MSW (57.2% [274/479]), and women (63.5% [442/696]). The percentage employed was: MSM (71.9% [732/1018]), MSW (70.2% [336/479]), and women (67.7% [471/696]). In high-income countries, the median duration of HIV reported was: 9, 4, and 5 years for MSM, MSW, and women, respectively. In middle-income countries, the median duration was 3, 6, and 6 years for MSM, MSW, and women, respectively.
Differential prevalence and trends in self-reported health outcomes
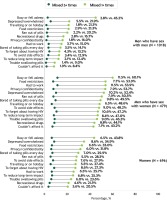
Differential trends in self-reported diagnosis of non-HIV conditions were observed among MSM, MSW, and women, by time since HIV diagnosis (Table 1). Among MSM and MSW, self-reported diagnosis of anemia did not change significantly by duration of HIV; among women however, the percentage reporting anemia increased from 13.5% (44/325) for 0–4 years to 28.3% (13/46) for ≥30 years since HIV diagnosis (p-trend <0.001). Prevalence of self-reported diagnosis of insomnia increased significantly among MSM (AAPC=23.0, p<0.001) and women (AAPC=28.3, p<0.001) but did not change significantly among MSW. Self-reported diagnosis of kidney, bone, and liver conditions, as well as mental illness and substance-use disorder, increased significantly with increasing HIV duration among all groups (all age-adjusted p-trend <0.05) (Table 1). Overall, 24.2% [116/479] of MSW reported suboptimal health on all four domains assessed (sexual/mental/physical/overall), significantly higher compared to MSM (18.0% [183/1018], p=0.005), but not significantly different from women (22.8% [159/696], p=0.585). Examination of the individual health domains showed that, compared to MSM, MSW reported significantly lower prevalence of optimal physical (51.6% [247/479] vs 67.0% [682/1018]), mental (45.3% [217/479] vs 62.6% [637/1018]), and overall health (52.6% [252/479] vs 63.6% [647/1018]), as well as viral suppression (57.0% [273/479] vs 89.2% [908/1018]) (all p<0.001). Women reported higher prevalence of optimal mental health (58.2% [405/696], p<0.001) and viral suppression (62.8% [437/696], p=0.046) than MSW as well. Difficulty swallowing pills among MSW (56.8% [272/479] was significantly higher compared to MSM (20.2% [206/1018]) and women (36.5% [254/696]) (all p<0.001). The median number of reasons reported for missing HIV medication ≥1 time in the past month was 7, 2, and 1 for MSW, women, and MSM, respectively, in the overall population, and 9, 4, and 3, respectively, among those who missed HIV medication ≥1 time in the past month. More MSW missed HIV medication for each of the assessed reasons than MSM or women (Figure 1).
Table 1
Trends in self-reported diagnosis of selected outcomes by time (years) since HIV diagnosis, among men who have sex with men (N=1018), men who have sex with women (N=479), and women (N=696) in 25 countries, 2019
| Condition | Group | 0–4 % | 5–9 % | 10–14 % | 15–19 % | 20–24 % | 25–29 % | ≥30 % | AAPC (unadjusted)a | Age adjusted p-trendb |
|---|---|---|---|---|---|---|---|---|---|---|
| Anemia | MSM | 6.9 | 8.1 | 5.8 | 9.1 | 9.2 | 6.7 | 11.4 | 6.5 (-1.5 to 15.1) | 0.745 |
| MSW | 6.9 | 12.7 | 5.7 | 5.0 | 12.5 | 36.4 | 7.1 | 22.7 (-6.0 to 60.1) | 0.214 | |
| Women | 13.5 | 22.3 | 16.4 | 17.1 | 19.6 | 45.2 | 28.3 | 17.6 (3.2 to 34.0) | <0.001 | |
| Bone disease | MSM | 1.7 | 1.9 | 8.0 | 10.9 | 14.5 | 10.0 | 27.8 | 49.9 (25.0 to 79.7) | <0.001 |
| MSW | 2.7 | 3.9 | 2.9 | 2.5 | 6.3 | 18.2 | 42.9 | 56.0 (25.5 to 94.0) | 0.007 | |
| Women | 3.1 | 3.2 | 2.7 | 11.4 | 15.2 | 26.2 | 39.1 | 56.4 (42.7 to 71.3) | <0.001 | |
| Insomnia | MSM | 9.2 | 7.6 | 21.9 | 26.4 | 19.7 | 28.3 | 30.4 | 23.0 (7.2 to 41.2) | <0.001 |
| MSW | 8.4 | 9.8 | 8.6 | 20.0 | 12.5 | 18.2 | 7.1 | 16.3 (-2.1 to 38.0) | 0.869 | |
| Women | 8.0 | 14.9 | 20.5 | 18.6 | 23.9 | 28.6 | 43.5 | 28.3 (18.6 to 38.8) | <0.001 | |
| Kidney disease | MSM | 0.9 | 1.9 | 1.5 | 1.8 | 6.6 | 8.3 | 16.5 | 62.3 (42.9 to 84.4) | 0.002 |
| MSW | 3.1 | 3.9 | 5.7 | 7.5 | 12.5 | 27.3 | 7.1 | 41.8 (18.5 to 69.7) | 0.036 | |
| Women | 2.8 | 4.3 | 5.5 | 4.3 | 8.7 | 9.5 | 6.5 | 21.2 (7.9 to 36.0) | 0.025 | |
| Liver disease | MSM | 4.9 | 9.0 | 10.2 | 10.9 | 15.8 | 18.3 | 21.5 | 24.7 (16.6 to 33.4) | 0.013 |
| MSW | 6.1 | 3.9 | 2.9 | 12.5 | 37.5 | 54.5 | 57.1 | 52.7 (30.2 to 79.0) | <0.001 | |
| Women | 4.0 | 6.4 | 11.0 | 11.4 | 17.4 | 28.6 | 52.2 | 52.4 (43.5 to 61.9) | <0.001 | |
| Mental conditions | MSM | 17.1 | 22.4 | 33.6 | 40.9 | 26.3 | 36.7 | 35.4 | 12.9 (-0.6 to 28.3) | 0.001 |
| MSW | 5.4 | 8.8 | 2.9 | 7.5 | 25.0 | 27.3 | 14.3 | 29.9 (6.5 to 58.4) | 0.004 | |
| Women | 8.3 | 19.1 | 24.7 | 22.9 | 30.4 | 31.0 | 41.3 | 25.0 (11.2 to 40.5) | <0.001 | |
| Substance misuse | MSM | 4.9 | 5.2 | 13.9 | 10.0 | 10.5 | 8.3 | 13.9 | 16.0 (-3.4 to 39.4) | <0.001 |
| MSW | 4.2 | 5.9 | 2.9 | 15.0 | 37.5 | 45.5 | 35.7 | 54.3 (29.7 to 83.6) | <0.001 | |
| Women | 4.3 | 4.3 | 12.3 | 17.1 | 15.2 | 14.3 | 19.6 | 27.4 (0.8 to 60.9) | <0.001 |
a To test for trends, joinpoint regression analyses were performed. For each line segment in the joinpoint regression, we used NCI’s Joinpoint 4.0.1 software to compute annual percentage changes (APCs) with 95% confidence intervals; average annual percentage changes (AAPC) were computed to summarize overall trends across all segments. To allow robust estimates, duration of HIV was coded in intervals of 0–4, 5–9, 10–14, 15–19, 20–24, 25–29, and ≥30 years.
Figure 1
Percentage who reported missing antiretroviral treatment 1+ or 5+ times within the past month, by reason reported among men who have sex with men, men who have sex with women, and women
The percentage who reported awareness of the number of medicines in their HIV regimen was significantly lower among MSW (64.3% [308/479]) than either MSM (80.4% [818/1018], p< 0.001) or women (72.1% [502/696], p=0.004). The percentage of all surveyed MSW who considered efforts to minimize ART side effects as a priority when they first started treatment (44.7% [214/479]) was significantly lower compared to MSM (60.3% [614/1018]); also, more MSW (51.6% [247/479]) experienced side effects from their current HIV medication than MSM (35.4% [360/1018]) (all p<0.001). Differences with women were non-significant for both indicators above, related to side effects. Of those who experienced side effects, MSW (56.3% [139/247]) reported the highest percentage of those who missed HIV medication ≥1 time in the past month because of side effects, versus women (33.1% [113/341], p<0.001) or MSM (24.4% [88/360], p<0.001). Despite MSW reporting the lowest prevalence of any non-HIV comorbidities ‘ever been diagnosed with by a doctor or other healthcare professional’ (46.1% [221/479]) compared to either MSM (64.6% [658/1018]) or women (56.8% [395/696]) (all p<0.001), MSW reported a higher prevalence of polypharmacy (45.1%) compared to either MSM (38.5%, p=0.017) or women (38.2%, p=0.02) (Supplementary file Figure 1). The poorer results seen for MSW persisted even after adjusting for duration of HIV and various indicators of healthcare access.
Differences in perceived stigma, and attitudes towards sexual/reproductive health
Of those in a relationship within the pooled sample from middle- and high-income countries, MSW reported lower prevalence of sharing their HIV status with a spouse/significant other (70.1% [309/441]) compared to either MSM (79.0% [548/694], p=0.001) or women (75.5% [448/593], p=0.049). Overall, 48.9% [234/479] of MSW worried that taking their HIV pills every day increased the chance of sharing their HIV status with others, significantly higher than either MSM (29.1% [296/1018], p<0.001), or women (41.4% [288/696], p=0.011). The percentage that had shared their HIV status with only their primary HIV care providers but no other person, was higher among MSW (19.6% [94/479]) versus MSM (7.9% [80/1018], p<0.001), but did not differ significantly from women (15.7% [109/696], p=0.077). The percentage that had shared their HIV status with their family doctor not providing HIV care, among those with one, was lowest for MSW (50.3% [229/455]) versus MSM (76.3% [704/923], p<0.001) or women (66.7% [444/666], p<0.001). Within stratified analysis, 62.7% [79/126] of MSW in middle-income countries had ever refused to share their HIV status for fear of being seen differently versus 46.5% [164/353] of MSW in high-income countries (p=0.002).
Among all participants regardless of HIV duration, the percentage of all surveyed MSW who considered having children as a priority at the time of starting treatment (30.5% [146/479]) was 1.4 times higher compared with women (22.6% [157/696], p=0.002) but almost 6 times higher compared with MSM (5.4% [55/1018], p<0.001) (Table 2). Women in high-income countries were significantly less likely to consider having children as a priority at the time of starting treatment (19.2% [95/495]) compared to women in middle-income countries (30.8% [62/201], p=0.001); for MSW and MSM, no significant differences existed between middle- and high-income countries. Among the subset of the population who had been diagnosed for ≥1 year, changes in perceived priorities between the time of starting treatment versus the time of the survey differed by gender/sexual orientation and regionally (Supplementary file Table 1 and Supplementary file Figure 2). For example, the absolute change in perceived importance of preventing HIV transmission to a partner, when comparing time of starting treatment versus the time of the survey, respectively, was positive (i.e. an increase) among the following three groups: MSM in high-income countries (52.4% [432/825] vs 67.5% [557/825], p<0.001), MSW in middle-income countries (43.6% [51/117] vs 70.9% [83/117], p<0.001), and women in high-income countries (37.5% [173/461] vs 50.8% [234/461], p<0.001). Conversely, no change between the two time points was seen among MSM and women in middle-income countries, as well as MSW in high-income countries.
Table 2
Self-rated health and treatment experiences among men who have sex with men (N=1018), men who have sex with women (N=479), and women (N=696) living with HIV in 25 countries, 2019
| All men | MSM | MSW | All women | |||||
|---|---|---|---|---|---|---|---|---|
| HIC | MIC | HIC | MIC | HIC | MIC | HIC | MIC | |
| N=1301 | N=322 | N=859 | N=159 | N=353 | N=126 | N=495 | N=201 | |
| Self-rated health | ||||||||
| Self-reported viral suppressiona | 78.6 | 78.6 | 89.9 | 85.5 | 52.4 | 69.8 | 59.6 | 70.6 |
| Optimal physical healthb | 60.6 | 64.0 | 66.1 | 71.7 | 50.7 | 54.0 | 52.3 | 64.2 |
| Optimal mental healthb | 55.3 | 64.0 | 60.8 | 72.3 | 43.1 | 51.6 | 55.4 | 65.2 |
| Optimal sexual healthb | 46.0 | 57.1 | 47.3 | 64.2 | 44.2 | 52.4 | 46.9 | 53.7 |
| Optimal overall healthb | 58.5 | 62.7 | 61.9 | 72.3 | 53.3 | 50.8 | 50.3 | 62.2 |
| Reported polypharmacyc | 42.8 | 33.1 | 39.1 | 35.3 | 49.0 | 33.9 | 40.9 | 31.8 |
| Reported any non-HIV comorbidityd | 58.3 | 61.8 | 64.8 | 63.5 | 41.1 | 60.3 | 54.9 | 61.2 |
| Reported suboptimal treatment adherencee | 21.4 | 27.0 | 14.8 | 19.5 | 38.8 | 38.9 | 26.1 | 32.8 |
| Difficulty swallowing pillsf | 31.1 | 33.5 | 20.3 | 20.1 | 58.9 | 50.8 | 39.0 | 30.3 |
| Perceive that HIV has an overall negative impact on their life | 39.6 | 34.8 | 39.7 | 37.1 | 34.6 | 27.8 | 46.5 | 32.3 |
| Satisfied with current HIV medicationg | 73.4 | 65.2 | 79.2 | 69.2 | 60.9 | 58.7 | 63.6 | 66.2 |
| People with whom PLHIV have shared their HIV statush | ||||||||
| Spouse/significant other | 75.1 | 78.4 | 78.3 | 82.1 | 68.4 | 74.4 | 73.2 | 81.4 |
| Casual sex partners | 66.5 | 52.9 | 73.1 | 63.8 | 50.2 | 39.0 | 55.9 | 52.1 |
| Parents, siblings, and children | 65.5 | 62.6 | 67.4 | 60.5 | 59.8 | 62.1 | 73.3 | 79.8 |
| Close friends | 74.1 | 70.5 | 82.8 | 84.0 | 52.5 | 53.3 | 64.3 | 73.0 |
| Wider family/circle of friends | 51.0 | 35.3 | 58.9 | 37.2 | 32.9 | 32.2 | 41.9 | 46.8 |
| Family doctor not providing HIV care | 70.0 | 60.3 | 79.5 | 57.4 | 47.2 | 59.5 | 64.8 | 71.5 |
| Other HCPS not providing HIV care | 62.8 | 57.8 | 72.5 | 61.1 | 40.0 | 52.9 | 54.5 | 65.6 |
| Most of the people in my life | 30.5 | 20.1 | 35.2 | 21.7 | 20.4 | 20.2 | 28.2 | 35.7 |
| Co-workers | 35.9 | 28.8 | 40.0 | 30.2 | 28.4 | 28.1 | 30.5 | 41.5 |
| Reasons for which PLHIV ever refused to share their HIV status | ||||||||
| It has not been necessary/relevant to my daily interactions | 41.4 | 49.7 | 46.4 | 49.1 | 30.0 | 50.8 | 32.1 | 31.8 |
| I was worried that they would see or treat me differently | 57.8 | 64.6 | 62.5 | 67.3 | 46.5 | 62.7 | 54.5 | 61.7 |
| I was worried that they might then disclose my HIV status to others | 51.5 | 61.5 | 54.5 | 67.3 | 45.0 | 54.8 | 44.4 | 55.2 |
| I was worried that I might be excluded from activities | 35.1 | 47.2 | 34.6 | 48.4 | 37.4 | 46.8 | 43.6 | 31.3 |
| I was worried about being denied access to health care services | 16.1 | 19.9 | 14.9 | 20.1 | 20.4 | 19.0 | 21.4 | 21.9 |
| I was worried about being denied access to financial benefits/support | 19.2 | 14.6 | 18.2 | 12.6 | 21.8 | 15.1 | 18.2 | 13.9 |
| I was worried it might affect my friendships | 45.3 | 48.8 | 46.7 | 50.3 | 40.8 | 50.8 | 47.7 | 43.8 |
| I was worried I might lose my job | 30.8 | 38.2 | 31.5 | 47.2 | 29.2 | 32.5 | 42.0 | 30.3 |
| I was worried it might affect my romantic or sexual relationships | 39.4 | 46.3 | 45.8 | 47.8 | 25.5 | 43.7 | 25.9 | 36.8 |
| I was worried about my physical safety | 20.0 | 20.5 | 18.6 | 22.0 | 21.5 | 18.3 | 19.0 | 22.9 |
| I was worried about criminal prosecution | 11.6 | 8.1 | 8.8 | 6.9 | 19.5 | 7.9 | 10.1 | 5.0 |
| Treatment priorities at treatment initiationi | ||||||||
| To ensure side effects would be minimal | 55.3 | 57.1 | 59.7 | 63.5 | 43.1 | 49.2 | 47.1 | 51.2 |
| To ensure that the virus was suppressed enough so that I could not pass it on to a partner | 49.9 | 55.9 | 53.0 | 65.4 | 41.9 | 43.7 | 38.2 | 57.7 |
| To minimize the long-term impact of HIV treatment | 45.6 | 41.6 | 48.4 | 37.7 | 38.0 | 46.0 | 38.6 | 39.3 |
| To manage symptoms or illnesses caused by HIV | 50.7 | 56.2 | 53.6 | 53.5 | 42.5 | 61.1 | 47.9 | 55.7 |
| To keep the number of HIV medicines in my treatment to a minimum | 34.4 | 39.4 | 34.5 | 35.2 | 32.9 | 41.3 | 33.9 | 30.3 |
| To allow flexibility as to when I have to take the HIV medication (time of day, with or without food, etc.) | 35.5 | 40.4 | 37.3 | 44.0 | 31.2 | 34.9 | 28.9 | 40.3 |
| To ensure it was compatible with other medications/drugs/pills I was taking | 31.4 | 26.7 | 29.5 | 25.2 | 34.6 | 28.6 | 32.3 | 28.9 |
| That the treatment is available in my public health facility | 26.7 | 50.9 | 25.4 | 62.3 | 30.6 | 31.7 | 21.6 | 47.3 |
| The cost of the medication | 25.9 | 19.6 | 22.9 | 14.5 | 31.2 | 23.8 | 24.4 | 17.4 |
| To have the best option to allow me to have children | 12.3 | 20.5 | 5.1 | 6.9 | 28.0 | 37.3 | 19.2 | 30.8 |
MSM: men who have sex with men. MSW: men who have sex with women. HCP: healthcare provider. HIC: high-income country. MIC: middle-income country. Summed sample size for MSM and MSW is less than for all men (N=1623) because of missing/indeterminate information on sexual orientation. Countries were classified as high- or middle-income based on the World Bank classification that uses per capita gross domestic product. The middle-income countries were Argentina, Brazil, China, Mexico, Russia, and South Africa; all others were high-income countries.
a Viral suppression was defined as a response of ‘Undetectable’ or ‘Suppressed’ to the question ‘What is your most recent viral load?’.
b ‘Optimal health’ was assessed within the past four weeks; self-rating of health as ‘Good’ or ‘Very good’ was classified as ‘optimal’ (vs ‘Neither good nor poor’, ‘Poor’, or ‘Very poor’).
c Polypharmacy was defined as taking ≥5 pills/day (HIV and non-HIV conditions combined), or currently taking medicines for ≥5 medical conditions.
d Reported ‘ever been diagnosed’ with one of these conditions ‘by a doctor or other healthcare professional’: anemia, arthritis, bone disease, cancer, dementia, diabetes, gastrointestinal disease, heart disease, high cholesterol, hypertension, insomnia, kidney disease, lipodystrophy, liver disease, lung/respiratory disease, malabsorption, mental health disorder, neurological disorders, substance misuse, tuberculosis, or other non-HIV condition.
e Suboptimal adherence was defined as having missed HIV medication ≥5 times in the past month because of one or more reasons.
f Moderate to severe difficulty swallowing pills was self-reported and was defined as scores ≥3 (on an ordinal scale from 1 to 5) in response to the question: ‘In general, how easy or difficult do you find it to swallow pills?’.
g Respondents were classified as having treatment satisfaction if they scored ≥4 (on an ordinal scale from 1 to 5) in response to the question: ‘Overall, how satisfied are you with your current HIV medication?’.
h Sharing of HIV status within the various relationships was assessed with the question: ‘Other than your HIV healthcare providers, who knows about your HIV status?’. Within each of the assessed relationships, categorical response options were ‘Yes’, ‘No’, or ‘Not applicable’. The latter was excluded.
Among all MSM and women surveyed, those in high-income countries were more likely to be told of ‘Undetectable = Untransmittable’ by their HCP than their counterparts in middle-income countries (MSM: 73.1% [628/859] in high-income countries vs 56.6% [90/159] in middle-income countries, p<0.001; women: 68.9% [341/495] in high-income countries vs 54.2% [109/201] in middle-income countries, p<0.001). For MSW, the percentage who reported discussing ‘Undetectable = Untransmittable’ with their HCP was similar between high-income countries (57.5% [203/353]) and middle-income countries (57.9% [73/126], p=0.933) (Table 3). Women in middle-income countries were one of the groups to benefit the most from discussing ‘Undetectable = Untransmittable’ with their HCP; positive associations ranged from: feeling more ‘comfortable sharing’ their HIV status (AOR=2.89; 95% CI: 1.41–5.95); reduced fear of HIV-related discrimination in the workplace (AOR=0.46; 95% CI: 0.22–0.95) or in healthcare settings (AOR=0.59; 95% CI: 0.37–0.94); optimal sexual health (AOR=1.29; 95% CI: 1.04–1.60); belief that HIV treatment prevents transmission (AOR=5.18; 95% CI: 1.67–16.11); reduced odds of suboptimal adherence (AOR=0.34; 95% CI: 0.14–0.83); and increased odds of self-reported viral suppression (AOR=2.82: 95% CI: 1.20–6.64) (Table 4). The observed benefits of discussing ‘Undetectable = Untransmittable’ with HCPs among MSM in high-income countries included a combination of clinical and social outcomes, whereas for middle-income countries it was mostly social outcomes. For example, among MSM in high-income countries, discussing ‘Undetectable = Untransmittable’ with HCPs was associated with reduced odds of suboptimal adherence (AOR=0.55; 95% CI: 0.39–0.77), as well as higher odds of: belief that HIV medication prevented HIV transmission (AOR=2.22; 95% CI: 1.59–3.11); self-reported viral suppression (AOR=2.37; 95% CI: 1.42–3.97), optimal sexual health (AOR=1.52; 95% CI: 1.01–2.32), and sharing of HIV status with a spouse/partner (AOR=2.40; 95% CI: 1.42–4.07), close friends ( AOR=1.90; 95% CI: 1.17–3.08), wider circle of family/friends (AOR=1.55; 95% CI: 1.02–2.35), and ‘most of the people in my life’ (AOR=1.55; 95% CI: 1.25–1.93). Among MSM in middle-income countries, discussing ‘Undetectable = Untransmittable’ with HCPs was not significantly associated with treatment adherence or viral suppression, but was significantly associated with belief that ART prevented HIV transmission (AOR=2.47), optimal sexual health (AOR=2.89), and increased willingness to share HIV status with close family (AOR=1.50), wider circle of family/friends (AOR=1.74), family doctor (AOR=1.76), and coworkers (AOR=1.52), but not with a spouse/partner (AOR=0.44), the latter finding was seen among MSW in middle-income countries as well (all p<0.05) (Table 4). MSM in middle-income countries who reported discussing ‘Undetectable = Untransmittable’ with their HCP were less likely to withhold their HIV status in healthcare settings for fear of being denied treatment (AOR=0.20; 95% CI: 0.10–0.41).
Table 3
Relationship with healthcare providers among men who have sex with men (N=1018), men who have sex with women (N=479), and women (N=696) living with HIV in 25 countries, 2019
| All men | MSM | MSW | All women | |||||
|---|---|---|---|---|---|---|---|---|
| HIC | MIC | HIC | MIC | HIC | MIC | HIC | MIC | |
| N=1301 | N=322 | N=859 | N=159 | N=353 | N=126 | N=495 | N=201 | |
| Relationship with healthcare providersa | ||||||||
| I am given enough information to be involved in making choices about my HIV treatment | 66.4 | 50.6 | 72.6 | 49.7 | 50.7 | 54.0 | 64.4 | 47.3 |
| I feel I understand enough about my HIV treatment | 73.8 | 65.5 | 79.5 | 71.7 | 60.1 | 56.3 | 71.7 | 62.2 |
| HCP seeks my views about treatment before prescribing an HIV medication | 66.9 | 50.9 | 69.6 | 45.9 | 61.2 | 54.0 | 65.7 | 43.8 |
| HCP asks me if I have any concerns about the HIV medication I am currently taking | 66.5 | 61.8 | 68.2 | 62.3 | 62.6 | 63.5 | 66.9 | 52.2 |
| HCP tells me about new HIV treatment options that become available | 62.3 | 47.2 | 64.0 | 40.9 | 58.9 | 57.1 | 64.8 | 39.8 |
| HCP asks me frequently about any side effects I might be experiencing | 63.6 | 60.6 | 66.9 | 62.9 | 55.8 | 56.3 | 64.2 | 54.2 |
| HCP has told me about ‘undetectable = untransmissible’ (U = U) | 68.7 | 58.1 | 73.1 | 56.6 | 57.5 | 57.9 | 68.9 | 54.2 |
| I would like to be more involved when it comes to decisions about my HIV treatment | 59.0 | 73.0 | 59.4 | 74.8 | 55.8 | 72.2 | 66.5 | 82.1 |
| HCP meets my personal needs and considers the things that are important to me | 70.5 | 62.4 | 76.0 | 63.5 | 59.2 | 62.7 | 65.9 | 58.7 |
| No perceived barriers to discussing with HCP | 32.9 | 24.8 | 43.2 | 28.9 | 8.8 | 21.4 | 26.7 | 28.9 |
| Percentage comfortable discussing specific issues with HCPb | ||||||||
| The impact HIV is having on my life generally | 59.0 | 52.8 | 66.0 | 56.6 | 43.6 | 50.8 | 54.7 | 56.2 |
| Preventing HIV transmission | 61.6 | 60.9 | 70.8 | 67.9 | 39.4 | 52.4 | 56.0 | 63.2 |
| My emotional well-being | 55.7 | 52.8 | 64.0 | 56.6 | 37.1 | 49.2 | 49.3 | 56.7 |
| Privacy/confidentiality concerns | 55.0 | 49.4 | 62.2 | 49.1 | 39.1 | 50.8 | 47.1 | 54.2 |
| Having children | 44.7 | 44.4 | 47.1 | 45.9 | 38.5 | 42.1 | 45.3 | 50.7 |
| Illnesses caused by HIV | 62.0 | 53.1 | 69.8 | 50.9 | 42.8 | 52.4 | 54.1 | 60.2 |
| Side effects | 64.0 | 54.7 | 73.7 | 56.0 | 41.4 | 51.6 | 58.4 | 59.2 |
| Drug-drug interactions | 63.9 | 59.3 | 73.0 | 63.5 | 43.9 | 53.2 | 58.0 | 51.7 |
| Long-term effects (e.g. problems with bones, kidneys, liver) | 62.1 | 53.1 | 71.9 | 57.2 | 38.8 | 48.4 | 57.4 | 53.7 |
| Missing medication | 58.8 | 55.3 | 67.8 | 56.0 | 36.0 | 54.0 | 55.6 | 49.8 |
MSM: men who have sex with men. MSW: men who have sex with women. HCP: healthcare provider. HIC: high-income country. MIC: middle-income country. Summed sample size for MSM and MSW is less than for all men (N=1623) because of missing/indeterminate information on sexual orientation. Countries were classified as high- or middle-income based on the World Bank classification that uses per capita gross domestic product. The middle-income countries were Argentina, Brazil, China, Mexico, Russia, and South Africa; all others were high-income countries.
Table 4
Adjusted odds ratio with 95% CI of the relationship between a report of having discussed ‘Undetectable = Untransmittable’ with a healthcare provider, and various health-related outcomes among people living with HIV in 25 countries, 2019
[i] MSM: men who have sex with men. MSW: men who have sex with women. With each region, analyses were performed separately for MSM, MSW, and women. The primary exposure of interest was whether the participant had discussed ‘Undetectable = Untransmittable’ with their healthcare provider. Separate logistic regression models were fitted within the different strata, adjusted for employment, education, age, home ownership, disease duration, and country-specific human development index. Estimation of variance accounted for clustering of observations by country.
Differences in information seeking behavior and communication with healthcare providers
Among the subset of participants who reported having concerns about the long-term impact of their HIV treatment in the past 12 months, MSM, MSW, and women reported similar rates of educating themselves by ‘reading articles, forums, research or other information’ (MSM=48.1% [377/783]; MSW=50.0% [173/346]; and women=45.7% [241/527], p=0.446). Similarly, the percentage who reported ‘I have discussed my concern with my main HIV care provider’ was not significantly different by gender/sexual orientation (MSM=36.4% [285/783]; MSW=43.6% [151/346]; and women=40.0% [211/527], p=0.061). However, the percentage who indicated ‘I have talked to HIV patient support groups/organizations’ was significantly higher among MSW (41.9% [145/346]) and women (38.5% [203/527]) than MSM (26.2% [205/783]) (all p<0.001). Women in middle-income countries reported lower use of patient support groups/organizations than their counterparts in high-income countries (29.4% [50/170] vs 42.9% [153/357], p=0.003).
Pooled analyses of participants in middle- and high-income countries revealed that although desire to be involved in making treatment decisions was no different between MSW (60.1% [288/479]) and MSM (61.8% [629/1018], p=0.538), significantly fewer MSW reported being involved in their HIV care compared to MSM. For example, a significantly lower percentage of MSW reported the following indicators, compared to MSM, respectively: ‘I am given enough information to be involved in making choices about my HIV treatment’ (51.6% [247/479] vs 69.1% [703/1018], p<0.001), ‘My provider seeks my views about treatment before prescribing an HIV medication’ (59.3% [284/479] vs 65.9% [671/1018], p=0.013), or ‘My provider asks me frequently about any side effects I might be experiencing with my HIV treatment’ (56.0% [268/479] vs 66.3% [675/1018], p<0.001). MSW had the lowest percentage of those with no perceived barriers to discussing health issues with their HCP (12.1% [58/479]) vs MSM (41.0% [417/1018], p<0.001) or women (27.3% [190/696], p<0.001). Of surveyed PLHIV who ever wanted a new medication different from the one they were on and who discussed with their HCP, a significantly higher proportion of MSW received the medication (80.1% [213/266]), compared to MSM (54.3% [191/352], p<0.001), or women (62.6% [174/278], p<0.001). Of those who were not prescribed the medication after discussing with their HCP, only two reasons differed significantly by gender/sexual orientation, both of which were highest among MSW: medication not available (MSW=39.6% [21/53], MSM=24.2% [39/161], and women=22.1% [23/104], p=0.046), and medication not covered by insurance (MSW=20.8% [11/53], MSM=5.6% [9/161], and women=9.6% [10/104], p=0.005). MSW overall reported less satisfaction with their current HIV medication than MSM (60.3% [289/479] vs 77.6% [790/1018], respectively, p<0.001); they were also less satisfied with the care they received from HCPs in relation to whether it met their personal needs (60.1% [288/479] vs 74.1% [754/1018], p<0.001). Furthermore, MSW were more likely than MSM to perceive room for improvement with their HIV medication (43.4% [208/479] vs 33.0% [336/1018], p<0.001). None of these indicators differed significantly between MSW and women.
DISCUSSION
We found significant differences among MSM, MSW, and women, in relation to HIV duration, treatment needs, perceived stigma, and self-efficacy in discussing salient issues with HCPs. MSW had poorer health outcomes in general, especially when compared to MSM. This is a new finding that adds to the body of global HIV research; many global studies we reviewed did not examine MSW specifically15-20. Our results also showed that women presented needs that were not dissimilar to MSW in many respects. MSM appeared to be the most educated about HIV at the time of initiating ART, as they reported the highest percentage of those who prioritized key issues at that time of starting treatment. Furthermore, MSM were currently more health-literate and engaged, as evidenced by greater awareness of the number of medicines in their treatment, greater perceived comfort in discussing treatment challenges with HCPs, and greater overall involvement in care. Not surprisingly, MSM reported the highest prevalence of treatment adherence and virologic suppression. Efforts to narrow disparities by gender/sexual orientation must consider the overlapping and unique needs of MSW and women, and seek ways to address them. There is need to expand gender focus of public health programs and recognize the unmet and evolving needs of other groups. For example, in the Healthy People 2020 targets for HIV in the United States, targets specific to MSM exist (e.g. HIV-14.2 ‘Increase the proportion of men who have sex with men (MSM) who report having been tested for HIV in the past 12 months’)21. Lessons learned from such targeted interventions among MSM could be applied to groups that are currently disadvantaged, including MSW and women.
Despite a lower prevalence of non-HIV comorbidities, MSW reported poorer overall health, greater polypharmacy, and poorer virologic control. The report of fewer comorbidities ‘ever been diagnosed with by a doctor or other healthcare professional’ may possibly signal underdiagnosis, especially as studies have suggested higher rates of non-retention in care, having no access to care, or being un-insured among MSW22,23. Furthermore, MSW in our study were the least likely to share their HIV status with HCPs who were not their main HIV care providers, which may potentially contribute to fragmented care. However, even when engaging with their main HIV care providers, our findings indicated several communication challenges of a two-way nature between MSW and their providers. For example, despite having a higher prevalence of side effects from HIV medications than MSM, MSW were less comfortable than MSM to discuss side effects with their HCPs; at the same time, more MSM than MSW reported that their HCPs frequently asked them about any ART side effects they may be experiencing. Poor adherence among MSW was multifactorial, emphasizing that treatment planning for MSW can only be optimized if lifestyle factors that disrupt adherence are carefully considered. HCPs need to have more open conversations with MSW about treatment challenges and alternatives, as well as the implication of poor adherence for transmission risk.
Our results further showed that MSW, despite deeming the prevention of onward HIV transmission as one of their top three treatment priorities, had the lowest prevalence of sharing their HIV status with their sexual partners. Furthermore, even though MSW prioritized having children as a key treatment goal to a greater extent than MSM and even women, over one-third of MSW did not believe that ART prevented disease transmission, while over 2 in 5 reported not discussing ‘Undetectable = Untransmittable’ with their HCP. Disease transmission among MSW and women are deeply intertwined as women are more likely to be infected through heterosexual contact24,25. Differential peaks in age-specific HIV diagnosis rates have been noted between European males and females: rates were highest in men aged 25–29 years, versus the 30–39 years group for women24. Being diagnosed with HIV during those ‘prime’ years in a woman’s life may have profound implications socially, professionally, physically, sexually, and emotionally26-29. Many women living with HIV first learn about their HIV diagnosis during pregnancy, during which time treatment adherence may be particularly challenging5. A holistic consideration of psychosocial and emotional outcomes, beyond virologic control, may improve quality of life as espoused in the fourth ‘90’ target and accelerate progress towards other national or international targets30-32.
Our findings show that progress towards the UNAIDS 9090-90 targets may vary among subgroups. For MSW who reported poor rates of treatment adherence and viral control, as well as poorer self-efficacy in communicating treatment challenges with HCPs, there may be challenges attaining the 3rd 90 target (viral suppression). For women in high-income countries, where the data suggested later age at diagnosis, there could be challenges with the 1st 90 target (diagnosis). Older age at diagnosis could mean either later infection in life, or a lag period between infection and diagnosis. The basis for concern about increased risk of HIV infection among older women is from research findings indicating decreased condom use among women in older age, possibly from the misperception that protection is no longer necessary as pregnancy may no longer be a concern during menopause33. The array of differences identified in our study by gender/sexual orientation should be considered when planning or implementing interventions aimed at enhancing HIV care or prevention at all levels, including primary (preventing onward transmission), secondary (HIV screening among high-risk groups), and tertiary (HIV treatment and enhancing quality of life among PLHIV).
Strengths and limitations
The main strengths of this study are the use of a large international sample of persons living with HIV to examine gender differences in evolution of patients’ preferences in HIV care and perceived treatment needs. Yet, limitations exist. First, because of sample size limitations, we could not examine certain categories of gender/sexual orientation, including individuals identifying as bisexual, asexual, pansexual, or other identities. Furthermore, whereas men were analyzed in two separate categories of MSM and MSW, all women were analyzed in one category regardless of sexual orientation because of small sample size for women who have sex with women. Second, only associations can be drawn from the cross-sectional design. Third, the nonprobabilistic sampling may limit generalizability. Finally, cultural/regional differences in openness regarding discussing issues of sexuality may introduce some social/cognitive biases for questions deemed to be of a personal nature.
CONCLUSIONS
Among respondents in this international survey, key differences by gender and sexual orientation were observed in disease onset, treatment needs, and aspirations. MSW in general had poorer health-related outcomes. More differences existed between MSW and MSM, than between MSW and women for several health-related outcomes. Furthermore, where differences existed, the MSW-women gap was smaller than the MSW-MSM gap. These disparities underscore why a one-size-fits-all approach cannot yield optimal outcomes in patient care. Consideration should be given to patient concerns to help tailor treatment in a manner that improves their health-related quality of life.


